A synthetic biology design is not useful simply because it exists on a screen. It has to reach a biological system in a form the system can read, maintain, and express. A gene circuit, pathway, sensor, or protein design may look precise in software, but the living result depends on where that DNA goes, how it is carried, how many copies are present, how stable the carrier is, and how the host cell responds.
That is the world of plasmids, vectors, and delivery. The words sound like technical background, but they shape many of the practical questions in synthetic biology. The same sequence can behave differently when it rides on a small circular DNA molecule, sits inside a chromosome, appears briefly for a test, or is delivered to a cell type that handles foreign DNA poorly. The design is only one part of the story. The biological address matters.
If DNA Synthesis and Assembly explains how a designed sequence becomes a physical construct, this guide follows the next question. How does that construct enter a useful biological context without losing the meaning of the design?
A Vector Is More Than a Container
In ordinary language, a vector is a carrier. In synthetic biology, it is the DNA vehicle or delivery system that helps move a designed sequence into a host or biological platform. A vector might be a plasmid, a viral delivery system in appropriate research or therapeutic contexts, a chromosome-integration construct, or another engineered format designed for a particular organism and use.
It is tempting to picture the vector as a neutral box that holds the important cargo. That picture is too simple. The carrier affects the cargo. A vector can influence how strongly a gene is expressed, how long the design persists, how many copies exist in each cell, which organisms can maintain it, how easy it is to verify, and what safety questions follow. The same coding sequence can be quiet in one context, burdensome in another, unstable in a third, and useful in a fourth.
This is why synthetic biology cannot treat delivery as a housekeeping step after the interesting design work is complete. The vector helps define the experiment. It decides whether the cell sees the design as a temporary visitor, a maintained accessory, or part of its inherited genome. Those states lead to different kinds of evidence.
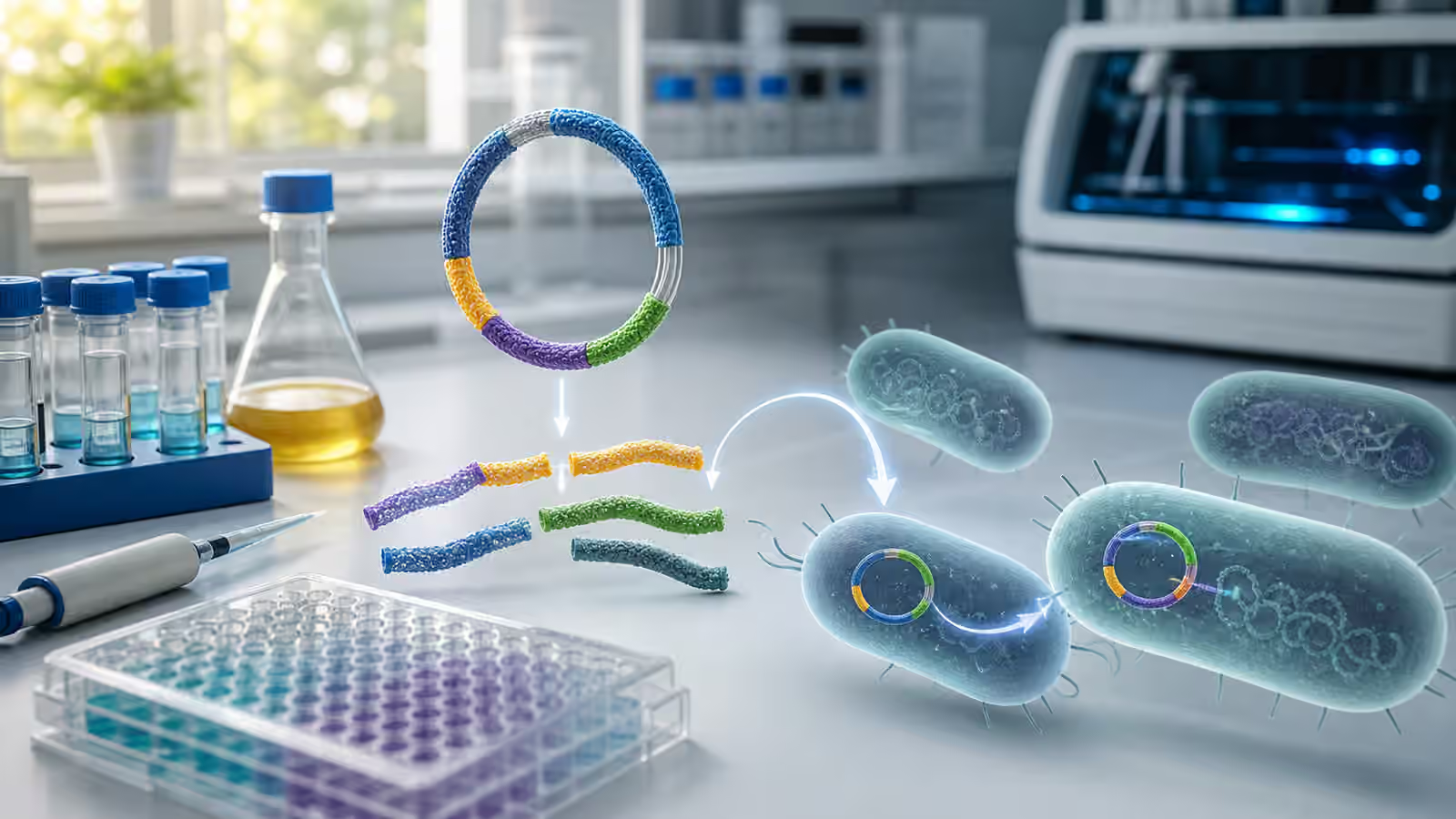
Plasmids Make Testing Faster
Plasmids are circular DNA molecules that can be maintained inside many microbes and some other biological systems outside the main chromosome. They became central to biotechnology because they make certain kinds of design work easier to test. A plasmid can carry a gene, regulatory elements, a reporter, a pathway segment, or a small circuit. It can be assembled, verified, introduced into a compatible host, and compared against alternatives.
That makes plasmids attractive for early experiments. A team can test several promoter strengths, enzyme variants, sensor outputs, or pathway arrangements without rebuilding the host genome every time. Plasmids fit naturally into the design-build-test-learn rhythm described in Biofoundries Explained , where measured iteration matters more than one perfect design.
But plasmids also bring tradeoffs. A plasmid is something the cell has to copy and maintain. That can create burden. If the plasmid asks the cell to make a costly protein or run a demanding pathway, cells that lose the plasmid or weaken the design may grow faster. Over time, the population can drift away from the intended engineered state. A short experiment may look promising while a longer process reveals instability.
Copy number is another quiet design variable. Some plasmids are present in many copies per cell, while others are maintained at lower copy number. More copies can increase expression, but more expression is not automatically better. It can overload folding machinery, drain resources, create toxic intermediates, or make the cell grow slowly. Lower copy number can be gentler and more stable, but may produce a weaker signal or less product. The plasmid is therefore part of the tuning system, not merely a storage device.
Genome Integration Changes the Promise
Sometimes the goal is not to maintain a separate plasmid. The design may need to become part of the host genome. Genome integration can make an engineered function more stable because the DNA is inherited as part of the chromosome rather than carried as an extra element. For production strains, long experiments, repeated passages, or workflows where selective pressure is undesirable or impractical, that stability can matter.
Integration also changes the design problem. A chromosomal insertion usually exists at lower copy number than a high-copy plasmid. It may produce less expression unless the surrounding regulatory design compensates. The insertion site can affect behavior because genomes are not uniform landscapes. Some regions are more active, more stable, or less disruptive than others. A change that is stable may also be harder to reverse, harder to retune, and more dependent on how the host manages its own DNA.
The existing guide to Genome Editing in Synthetic Biology follows that broader editing story. For plasmids and delivery, the lesson is narrower: stability is not free. A design can be easier to maintain once integrated, but the act of placing it into the genome adds its own verification, measurement, and safety burden. A stable wrong design is still wrong. A stable burdensome design can still be selected against if cells find ways to silence or damage it.
This is one reason Strain Engineering is such a careful craft. A production cell is not built by choosing plasmid or chromosome in isolation. The whole living system has to balance growth, pathway activity, product tolerance, genetic stability, process conditions, and measurement.
Delivery Depends on the Host
The phrase getting DNA into cells hides a large amount of biological variety. Bacteria, yeast, mammalian cells, plant cells, algae, fungi, and cell-free systems do not accept and use DNA in the same way. Some hosts are familiar laboratory workhorses with well-established tools. Others are attractive because of what they can make or tolerate, but harder to engineer. A host that looks perfect for a product may be frustrating because delivery is inefficient, unstable, or poorly understood.
Responsible educational writing should not turn delivery into a protocol. The evergreen point is that delivery is host-specific. The cell envelope, cell wall, membrane behavior, DNA repair systems, immune-like defenses, growth conditions, and stress responses all influence what happens to introduced genetic material. In some organisms, a construct may enter easily but express poorly. In others, it may enter rarely but perform well once established. In still others, the host may degrade, silence, rearrange, or reject the design.
The delivery method also shapes what kind of result can be trusted. A transient expression test may show that a protein can be made briefly, but it does not prove that a stable production strain exists. A mixed population may contain cells with different constructs, copy numbers, or integration events. A successful-looking sample may reflect the small fraction of cells that received the design rather than the whole culture. Without careful verification, delivery can create ambiguity that later measurements cannot fix.
Temporary Expression Has Its Own Uses
Not every synthetic biology experiment needs a durable change. Temporary expression can be useful when researchers want to test whether a design is readable, whether a protein can be produced, whether a reporter gives a signal, or whether a cell responds in a short window. In some research settings, transient systems can speed learning because they avoid the slower work of establishing stable lines or strains.
Temporary expression is not lesser work, but it answers a different question. It can show that a design has activity under a particular condition. It may not show that the design will remain stable, behave predictably over generations, or fit a manufacturing process. A result can be real and still narrow.
Cell-free systems change the question again. In Cell-Free Synthetic Biology , the design does not have to enter a living cell at all. The DNA or RNA is used by biological machinery outside a whole organism. That can make prototyping, education, sensing, or specialized production easier in some contexts, while removing other cellular complications. Yet the construct still needs the right format, the right regulatory elements, and appropriate controls. Delivery disappears as a cellular barrier, but compatibility remains.
Verification Connects the Carrier to the Claim
A guidebook about vectors can sound like a guidebook about objects: plasmids, backbones, insertion sites, delivery systems, and host cells. The deeper issue is evidence. When someone claims that a synthetic biology design worked, the reader should be able to ask what actually entered the system and what form it took.
Was the construct sequence verified? Was the intended plasmid present? Was the copy number or integration state understood well enough for the claim? Was the population uniform or mixed? Did the cells maintain the design over the relevant time? Was the output compared against controls that separate the intended function from background behavior?
Those questions connect directly to Biological Measurement and Controls . A bright signal, a product peak, or a growth change means less if the identity of the genetic material is uncertain. The vector is part of the measurement chain. If the delivery step is messy, the final data may be hard to interpret even when the instrument is working properly.
Good verification also protects design learning. A failed experiment can teach something useful if the team knows what was built and where it went. It teaches much less if the construct was wrong, the host carried a mixture, or the design disappeared before the assay. Synthetic biology advances through cycles of interpretation, and interpretation depends on knowing the physical state of the system.
Safety Belongs at the Delivery Layer
Vectors and delivery systems carry safety meaning because they influence persistence, host range, access, and movement. A construct intended for a contained microbial test is not the same as a construct intended for a mammalian cell study, a plant system, a therapeutic research setting, or an environmental proposal. The relevant safeguards depend on the organism, the genetic change, the carrier, the product, the scale, and the context.
The guide to Synthetic Biology Safety separates biosafety and biosecurity in more detail. At the delivery layer, the practical habit is to ask where the design can go, how long it can persist, who can access it, what happens if it reaches the wrong context, and what documentation follows it. A vector can make a design easier to test, but ease of movement is exactly why access control, containment, screening, labeling, and recordkeeping matter.
Safety thinking should also avoid exaggeration. Many plasmid-based experiments are routine in trained, contained settings. Many delivery systems are narrow tools for specific hosts. The point is not to make every carrier sound alarming. The point is to avoid pretending that the form of delivery is irrelevant. Biology reads context, and safety systems have to read it too.
The Address Shapes the Biology
Synthetic biology often begins with the romance of design: a sequence, a circuit, a pathway, a protein, a sensor, a possibility. Plasmids, vectors, and delivery bring that romance down to the practical question of address. Where will the design live? How will it arrive? How many copies will the host carry? How long will the design persist? How will the result be verified? What kind of evidence will that placement support?
Those questions explain why two projects with similar DNA can have different outcomes. One uses a plasmid for fast screening. Another integrates a pathway into a chromosome for stability. Another uses transient expression to answer an early question. Another avoids living cells and tests the design in a cell-free platform. None of those choices is automatically best. Each answers a different version of what it means for a design to enter biology.
The most grounded view is that vectors are not just delivery hardware. They are part of the design itself. They shape expression, burden, stability, measurement, scale-up, and safety. A synthetic biology design becomes meaningful only when its sequence, carrier, host, process, and evidence all point to the same story.