Synthetic biology often becomes visible only after a cell changes behavior. A culture produces a color. A biosensor gives a signal. A microbe makes a protein, flavor molecule, pigment, or material precursor. By then, the story already has a result. The quieter middle step is the one that makes the result possible: a designed DNA sequence has to become a physical, verified construct that a biological system can actually use.
That step sounds almost administrative when reduced to a phrase like DNA synthesis and assembly. It is not. It is where a drawing on a screen begins to meet chemistry, handling, host biology, measurement, safety review, and the stubborn reality of living systems. A sequence file may look exact, but the laboratory question is more demanding. Is this the right sequence, in the right context, carried in the right form, entering the right system, and producing evidence that can be trusted?

If you have read Synthetic DNA Circuits , this guide sits just downstream of that design layer. A circuit explains what the DNA is meant to do. Synthesis and assembly explain how the intended instruction becomes a material object ready for testing.
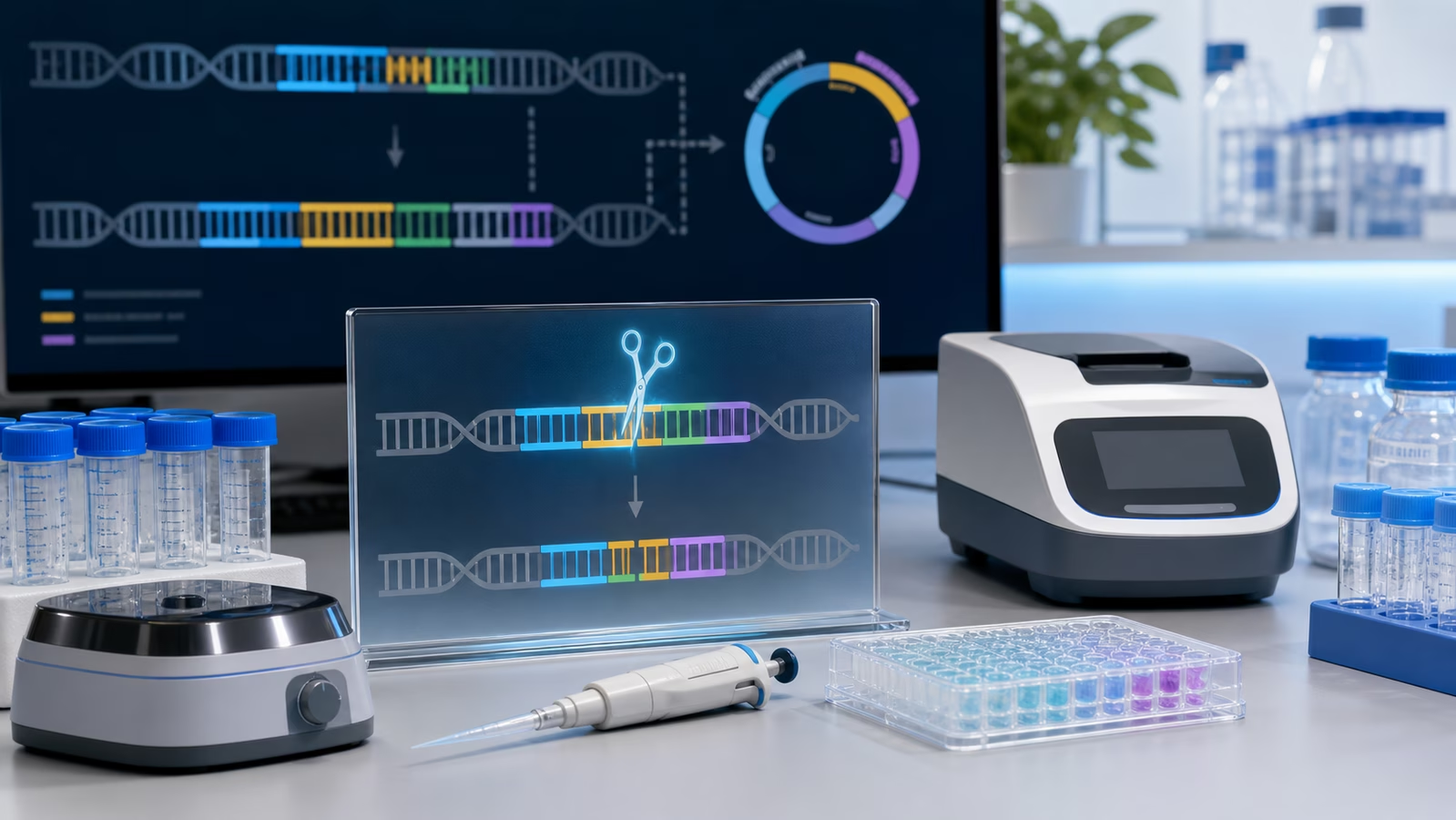
A Sequence Is a Hypothesis
The simplest public version of synthetic biology says that scientists write DNA. The phrase is useful, but it hides the amount of judgment involved before any DNA is made. A useful sequence is not just a string of A, C, G, and T. It may include genes, promoters, regulatory regions, terminators, binding sites, untranslated regions, tags, cloning scars, homology arms, selection features, and other context that affects how the final construct behaves.
Each part carries assumptions. A promoter may be chosen because it is expected to turn a gene on strongly, gently, or only under certain conditions. A coding sequence may be adjusted so the host can read it efficiently. A plasmid backbone may be selected because it has a compatible copy number, maintenance behavior, or history in a particular organism. A design meant for a bacterium may be unsuitable for yeast. A design that looks tidy in software may create unwanted repeats, unstable regions, difficult secondary structures, or burdensome expression once it enters a cell.
That is why a sequence is better understood as a hypothesis than as an answer. It says, in effect, if this DNA is built in this form and placed in this biological context, the system may behave in a useful way. The experiment begins by making that hypothesis physically testable.
What DNA Synthesis Changed
Older genetic engineering often depended more heavily on cutting, copying, and rearranging DNA that researchers already had in hand. Synthetic DNA changed the rhythm of the work. A team can now design a sequence digitally and order pieces of DNA that match the design, subject to provider constraints, safety screening, cost, length, complexity, and review. The result may be a short fragment, a longer gene, a library of variants, or a set of overlapping pieces intended for assembly.
This does not make biology instant. Synthesis providers still have to manufacture molecules, check quality, handle difficult sequences, and apply screening policies. Researchers still have to decide whether the sequence makes biological sense, whether it belongs in the chosen host, and whether the plan is appropriate for the intended setting. The ability to write DNA increases the importance of design discipline rather than removing it.
The biggest practical change is that researchers can explore more alternatives. Instead of testing only one natural gene, they may test several versions. Instead of accepting a pathway exactly as it appears in one organism, they may redesign expression levels, spacing, or enzyme variants. Instead of treating a circuit as a single precious object, they may build a family of related constructs and measure which one behaves best. That possibility is one reason Biofoundries Explained matters: modern synthetic biology is often a disciplined search across designs, not a single heroic edit.
Assembly Gives the Design a Body
DNA synthesis may provide the pieces, but assembly gives those pieces a usable form. A construct might be built as a plasmid that can be maintained inside a cell. It might be arranged for integration into a genome. It might be prepared for a cell-free system, where biological machinery operates outside a living cell. It might combine several DNA fragments that must sit in the correct order and orientation for the design to make sense.
The point is not the specific laboratory method. Methods change, and responsible work belongs inside trained settings with appropriate oversight. The evergreen idea is that assembly is about context. A gene by itself is rarely enough. The surrounding DNA influences whether the gene is copied, read, regulated, inherited, silenced, lost, or overexpressed. A beautifully designed enzyme sequence can disappoint if the construct places it under the wrong control. A biosensor can become noisy if its input and output pieces are poorly balanced. A production pathway can burden a host if every part is pushed too hard at once.
Good assembly thinking also respects future measurement. A construct should be easy enough to verify, compare, and document. If a team cannot tell which version was tested, or whether the parts were assembled as intended, the later biological result becomes ambiguous. The best construct is not only one that might work. It is one whose success or failure can teach the next design cycle something useful.
Verification Is Part of the Experiment
Once a construct exists, it has to be checked. Verification can sound like paperwork, but it is one of the main defenses against false confidence. DNA molecules are small, fragments can assemble incorrectly, cells can take up unexpected variants, and small sequence changes can matter. If the team tests the wrong construct, the result may look like biology when it is really a documentation or validation failure.
Verification asks whether the physical DNA matches the intended design closely enough for the experiment. It may involve sequence confirmation, size checks, orientation checks, comparison against the design file, and careful sample tracking. The details depend on the project, but the principle is stable: a biological claim rests partly on knowing what was actually built.
This connects directly to Biological Measurement and Controls . Controls are not only for final assays. They begin earlier, with reference materials, known constructs, careful metadata, and habits that make mistakes visible. A result from an unverified or poorly documented construct is weaker because the experiment cannot be interpreted cleanly. The cell may have answered a question, but the researcher may not know which question was asked.
The Construct Has to Live Somewhere
A construct is not complete simply because its DNA sequence is correct. It has to operate in a host or biological system. That host may be a bacterium, yeast, mammalian cell line, plant cell, cell-free extract, or another platform. Each setting changes what the construct means.
Inside a living cell, DNA competes for attention. The host has limited energy, ribosomes, polymerases, nutrients, cofactors, membrane capacity, folding machinery, and tolerance for stress. A construct that asks for too much can slow growth or select for cells that weaken or lose the design. A construct that looks stable in a short test may drift after many generations. A construct that makes a product in a tiny vessel may behave differently as oxygen transfer, mixing, pH, feeding, and waste handling change.
That is why synthesis and assembly belong beside Strain Engineering , not before it as a solved preliminary. A production strain is not just a host plus a sequence. It is a living system tuned so the construct, metabolism, regulation, process, and measurement can coexist long enough to make useful evidence or product.
Cell-free work changes the problem without eliminating it. Removing living cells can simplify some containment and toxicity questions, and it can make rapid prototyping easier. Yet the construct still has to be readable by the extract, compatible with the reaction conditions, and measured against appropriate controls. Cell-Free Synthetic Biology is a useful companion because it shows how the same designed DNA can behave differently when the living host is replaced by biochemical machinery.
Safety Starts Before the DNA Arrives
Safety is sometimes discussed as something that happens after an organism has been engineered. In responsible synthetic biology, it begins earlier. Before DNA is ordered or assembled, the team should understand what is being built, why it is needed, which organism or system will receive it, what hazards might be relevant, and what controls belong around the work.
DNA synthesis providers commonly apply screening and review processes, but provider screening is not a substitute for local responsibility. A harmless educational construct, a production enzyme, a diagnostic element, and a sequence connected to pathogenicity are not the same kind of object. The risk depends on sequence function, host, product, environment, scale, access, and intended use. Good practice also includes clear documentation, appropriate containment, careful waste handling, and avoiding casual movement of materials outside their approved context.
The guide to Synthetic Biology Safety covers biosafety and biosecurity more directly. The synthesis-and-assembly lesson is narrower: a sequence is already a material decision before it becomes a living test. Asking safety questions early is more practical than trying to retrofit them after the construct has become central to the project.
From Construct to Evidence
The finished construct is not the finish line. It is the beginning of evidence. Researchers still need to ask whether the construct produces the intended RNA, protein, signal, pathway behavior, material, or phenotype. They need to compare it with controls, repeat the test, notice byproducts, watch for instability, and decide what failure means. A construct can fail because the design idea was wrong. It can also fail because expression was poorly balanced, the host was unsuitable, the assay was misleading, or the process conditions exposed a hidden weakness.
This is why synthetic biology keeps returning to the design-build-test-learn loop. DNA synthesis and assembly sit in the build part, but they influence every other part. A design that is hard to assemble may slow learning. A construct that is hard to verify may weaken measurement. A poorly documented build may make good data unusable. A thoughtful assembly strategy can make the next round faster and more honest because it preserves the connection between the digital design, the physical DNA, and the biological result.
The most grounded way to think about DNA synthesis is not as a printer for life. It is a bridge. On one side is a designed sequence, clean and editable on a screen. On the other side is a biological system that can surprise, resist, adapt, and occasionally perform useful new work. The bridge has to carry more than letters. It has to carry context, verification, safety, and the discipline to learn from whatever the cell says back.